Pipeline 037
037
PROGRESS – Hit Generation
Novel targets to super-charge cytotoxic lymphocyte function
oNKo-037 is our most advanced small molecule program and it represents a novel immunotherapy target. Genetic manipulation of oNKo-037 enhances fitness of natural killer and T cells by increasing sensitivity to endogenous IL-15 cytokines.

PIPELINE 037
Systemic administration of cytokines can cause excessive activation of the immune system, resulting in adverse reactions and toxicity. To avoid this, we are homing in on targets that mediate the inhibitory effects of cytokine checkpoints to heat up immunologically ‘cold’ tumours.
By manipulating the response of cytotoxic lymphocytes like natural killer (NK) and T cells to low levels of endogenous cytokines, we can increase localized anti-tumour activity while reducing negative systemic effects.
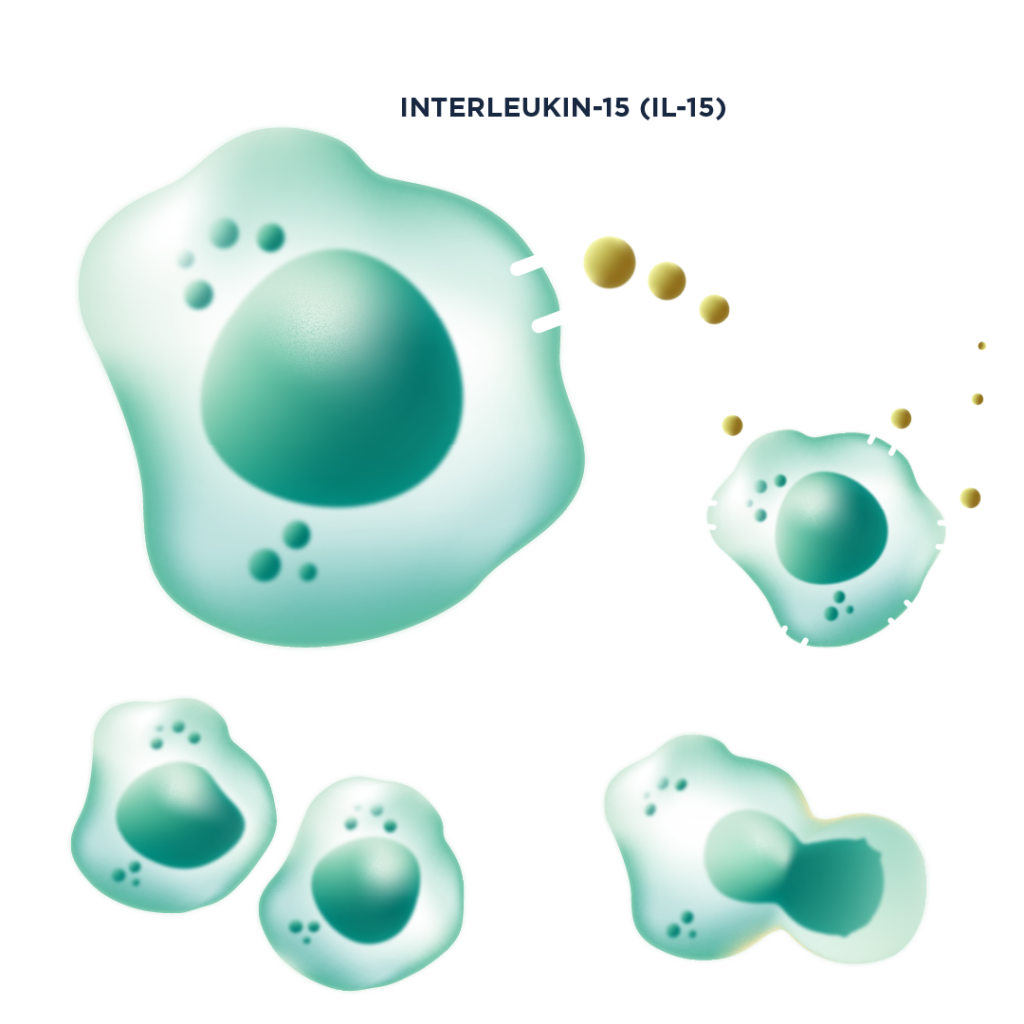
Using our CRISPR screening platform, we discovered that knockout of the oNKo-037 target significantly enhances NK cell sensitivity to the cytokine IL-15.
Functionally, oNKo-037 deletion drives enhanced fitness of both NK cells and T cells, including in the context of a chimeric antigen receptor (CAR). Under these conditions, the cells exhibit enhanced signalling, increased cellular metabolism, improved cytotoxicity, and elevated secretion of inflammatory cytokines.
oNKo-037 is our most advanced small molecule program and it represents a novel immunotherapy target. We have identified compounds that show binding to, and inhibition of, oNKo-037 without affecting closely related targets, and our team is currently exploring the structure-activity relationship for these compounds.